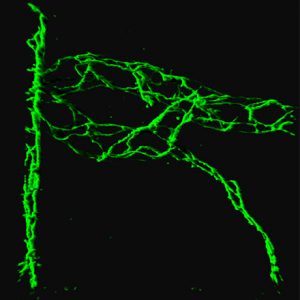
Blood brain barrier disruption and repair in mouse models of human disease
In CNS disorders including multiple sclerosis, stroke, cancer, and others, disruption of the blood-brain barrier (BBB) leads to tissue damage and deleterious clinical outcome by enabling infiltration of leukocytes and serum proteins. Barrier function is normally maintained by integrity of tight junctions (restricting paracellular permeability), suppression of caveolar transcytosis (transcellular permeability), and specific ion transporters. We have taken genetic, functional, and in vivo imaging approaches to uncover new aspects of barrier regulation, which have profound implications for disease and recovery, in the middle cerebral artery occlusion (MCAO) model of stroke and the experimental autoimmune encephalomyelitis (EAE) model of multiple sclerosis. Using in vivo two photon imaging in transgenic eGFP-Claudin5 mice with fluorescent tight junctions, we have identified dynamic BBB remodeling events that would be impossible to appreciate in fixed tissue preparations. We demonstrate regional anatomic differences in endothelial cell tight junction recycling, which may partially explain the enigmatic preferential vulnerability of the spinal cord to EAE. In MCAO, BBB breakdown occurs in two waves: first transcellular, then paracellular permeability. In EAE, this sequence is reversed. We find that remodeling of TJs precedes the onset of EAE clinical signs in mice and coincides with paracellular BBB leakage. In contrast, transcellular BBB leakage to albumin occurs later, at the peak of disease. Moreover, mice that lack caveolae have reduced EAE clinical severity. These findings suggest that TJ dissolution initiates disease onset, whereas caveolar transcytosis may enhance EAE severity. These findings also have relevance for BBB disruption in multiple neuroinflammatory states.
To establish novel methods to restore BBB integrity, we investigated the Wnt/β-catenin pathway, which is critical for BBB development. There exists an urgent need for new molecular strategies to protect and restore BBB function, either by strengthening tight junctions or suppressing trans-endothelial vesicular transcytosis. We have shown Wnt/β-catenin pathway upregulation in BBB endothelial cells in active lesions in MS brains and in the animal model experimental autoimmune encephalomyelitis (EAE) using detection of transcriptional targets Sox17 and Apcdd1 and the TCF/LEF1::H2B::eGFP β-catenin reporter mouse. We observed ligand dependent activation by neuronal expression of Wnt3 and ligand independent activation by liberation of β-catenin from adherens junctions. Genetic β-catenin loss of function in endothelial cells exacerbated EAE clinical presentation, demyelination, and CNS T cell infiltration. β-catenin loss of function enhanced endothelial expression of vascular cell adhesion molecule-1 (VCAM-1) and the transcytosis protein Caveolin-1, without disrupting tight junction proteins. These results suggest that re-activation of the developmental Wnt/β-catenin pathway in CNS endothelial cells is an endogenous BBB repair mechanism.
Inflammasome signaling contributes to EAE and seizure pathology
Pannexins are a class of gap junction proteins that, when open, permit transient passage of cytosolic contents such as ATP into the extracellular space down concentration gradients without necessarily causing cell death. I developed and employed a novel acute spinal cord slice preparation to demonstrate that ATP is released from EAE spinal cords in a Pannexin1-dependent manner. I showed that mice deficient in Pannexin1 channels exhibit delayed onset and decreased mortality of MOG peptide EAE, decreased demyelination and inflammation. A common clinical issue for MS patients is urinary incontinence, and the MOG EAE model also reproduces neurogenic bladder dysfunction, which we showed was ameliorated in Pannexin1-/- mice. In collaboration with colleagues, I showed that Pannexin1 deficient mice are protected from Kainic acid induced seizures. Pannexin1 channels both physically associate with components of the inflammasome and, by secretion of ATP, activate the inflammasome. My work demonstrates that this feedback loop is pathogenic in at least two neuroinflammatory disease models and can be arrested by genetically or pharmacologically targeting Pannexin1.
Glial cell connections by gap junctions are essential for myelination and brain homeostasis
Astrocytes and oligodendrocytes express a unique complement of gap junction proteins, involved in cell-to-cell transmission of signals. Previously it was unclear to what extent heterotypic cell pairs communicated by this mechanism. My work firmly established that astrocyte-oligodendrocyte gap junctional coupling is essential for normal development of myelin. In the absence of astrocyte gap junctions, oligodendrocytes make less myelin, fail to wrap axons, and undergo apoptosis. Astrocytes become hypertrophic and vacuolated. These mice exhibit behavioral abnormalities, but are not more susceptible to an animal model of multiple sclerosis. Other groups complemented this work by showing that mice deficient in oligodendrocyte gap junctions phenocopy mice deficient in astrocyte gap junctions. My work therefore supports and extends the previous hypothesis of an astrocytic syncytium, and established the critical importance of the astrocyte-oligodendrocyte pan-glial syncytium for white and gray matter ultrastructural and functional integrity.